Message from the CEO
“In June we announced that the first premature infant had been enrolled and dosed in the Company´s Phase ll clinical trial. This Phase ll trial is a randomized, double blind, parallel group, dose escalation, placebo-controlled multicenter study to investigate the safety and tolerability of IBP-9414 administered in preterm infants. The multicenter trial is being conducted in a number of neonatal intensive care units in the US and will enroll 120 premature infants in total. The first planned independent Data Safety Monitoring Board (DSMB) evaluation of safety data was performed on August 12th. The DSMB concluded that there were no objections to dose escalation based on the information provided to the DSMB.
We are very pleased that the trial has begun and is progressing according to plan. This is another major milestone in the development of a new pharmaceutical for this very sensitive group of patients.”
Staffan Strömberg,
Chief Executive Officer
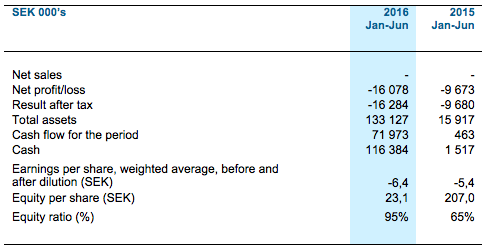
Financial summary
Significant events during the first six months
- ● Annual General Meeting decided on 8 February on repayment of conditional shareholder contributions by offsetting previously received group contributions by SEK 20.6m
- ● BioGaia AB (publ) distributed its entire holding (94.5 % of shares and 96 % of votes) in IBT to BioGaia’s shareholders
- ● IBT’s shares were listed on Nasdaq First North
- ● IBT completed a guaranteed share issue which generated approximately SEK 89m after deduction of issue costs
- ● The first premature infant has been enrolled and dosed in the Company’s Phase ll clinical trial
Significant events after the reporting period
- ● There were no significant events after the reporting period prior to the date of publication of this interim report
For additional information please contact
Staffan Strömberg, CEO, phone: +46 8 410 145 55
Peter Rothschild, Chairman of the Board, phone: +46 8 410 145 55
Infant Bacterial Therapeutics AB
Bryggargatan 10
111 21 Stockholm
Phone: +46 8 410 145 55
Publication
The information in this Interim Report is such which IBT is obliged to make public pursuant to the EU Market Abuse Regulation and which is to be made public according to the Nasdaq regulations for companies listed on Nasdaq First North.
The information was submitted for publication, by the CEO stated above, at 08.00 a.m. CET on 19 August, 2016.
2016
Interim report January 1 – June 30 2016
Press Release + Interim report English version
Press release + Interim report Swedish version