Infant Bacterial Therapeutics’ Focus Areas

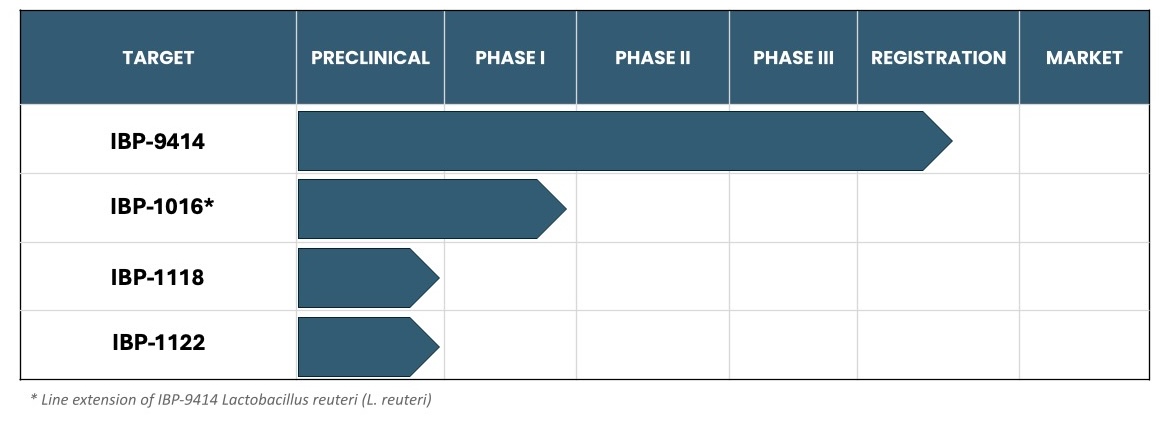
Product Pipeline
IBT’s current pipeline includes four development programs:
IBP-9414
Reduction of mortality among preterm infants
IBP-1016
Neonatal treatment of gastroschisis
IBP-1118
Prevention of retinopathy of prematurity (ROP)
IBP-1122
Prevention of antibiotic resistant hospital acquired infections
caused by vancomycin-resistant enterococci (VRE)
IBP-9414 is IBT’s lead drug candidate. The development plan for IBP-9414 consisted of two clinical studies: the safety and tolerance Phase II study and the pivotal Phase III study, “The Connection Study.”
In the Phase III study on premature infants(“The Connection Study”), which was completed in July 2024, the group treated with IBP-9414 showed a significant reduction in total mortality of 27% compared to the placebo group, meaning that widespread use of IBP-9414 could save more than 1,000 patients annually in the US alone. The treatment has been designated both a “Breakthrough Therapy” (March 2025) for gastrointestinal mortality and a “Rare Pediatric Disease,” reflecting its potential to meet a significant unmet medical need.

